Safety data
BLENREP had a well-established safety profile that was generally well tolerated
Most common adverse reactions (≥20%) in patients from DREAMM-7
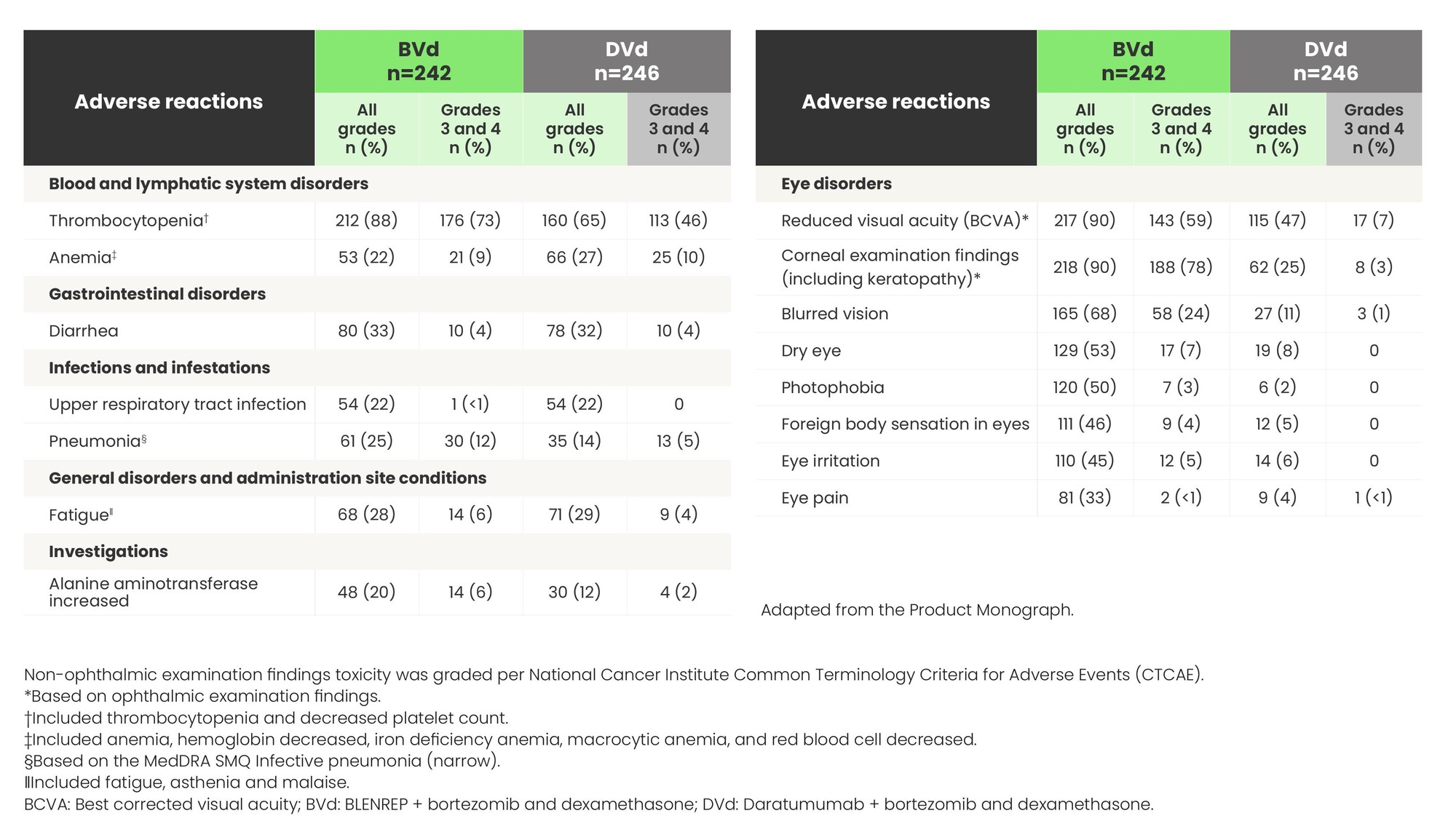
Non-ophthalmic examination findings toxicity was graded per National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE).
*Based on ophthalmic examination findings.
†Included thrombocytopenia and decreased platelet count.
‡Included anemia, hemoglobin decreased, iron deficiency anemia, macrocytic anemia, and red blood cell decreased.
§Based on the MedDRA SMQ Infective pneumonia (narrow).
‖Included fatigue, asthenia and malaise.
BCVA: Best corrected visual acuity; BVd: BLENREP + bortezomib and dexamethasone; DVd: Daratumumab + bortezomib and dexamethasone.
BLENREP had a well-established safety profile that was generally well tolerated
Most common adverse reactions (≥20%) in patients from DREAMM-8
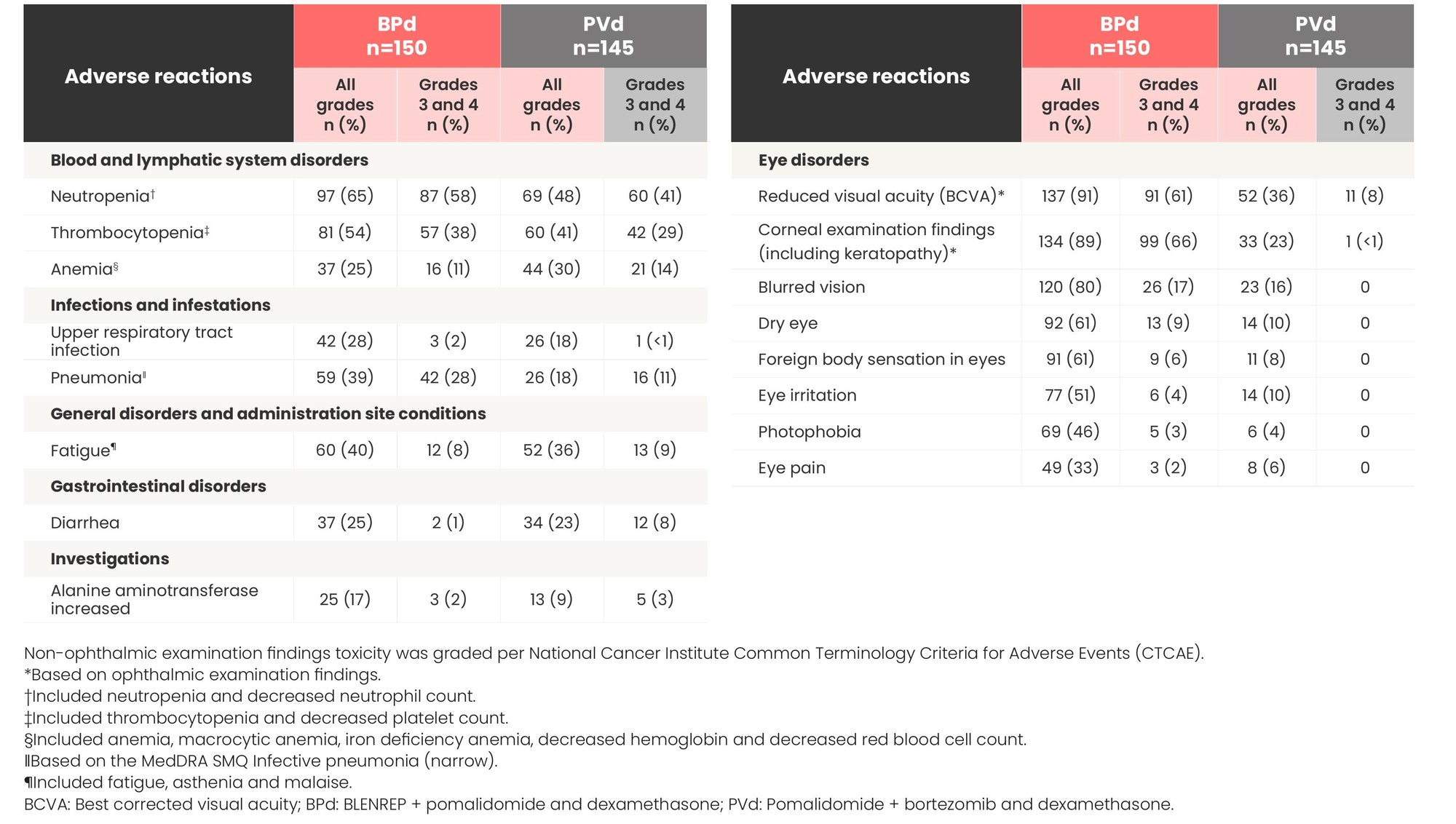
Non-ophthalmic examination findings toxicity was graded per National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE).
*Based on ophthalmic examination findings.
†Included neutropenia and decreased neutrophil count.
‡Included thrombocytopenia and decreased platelet count.
§Included anemia, macrocytic anemia, iron deficiency anemia, decreased hemoglobin and decreased red blood cell count.
‖Based on the MedDRA SMQ Infective pneumonia (narrow).
¶Included fatigue, asthenia and malaise.
BCVA: Best corrected visual acuity; BPd: BLENREP + pomalidomide and dexamethasone; PVd: Pomalidomide + bortezomib and dexamethasone.

