Study overview
Study design
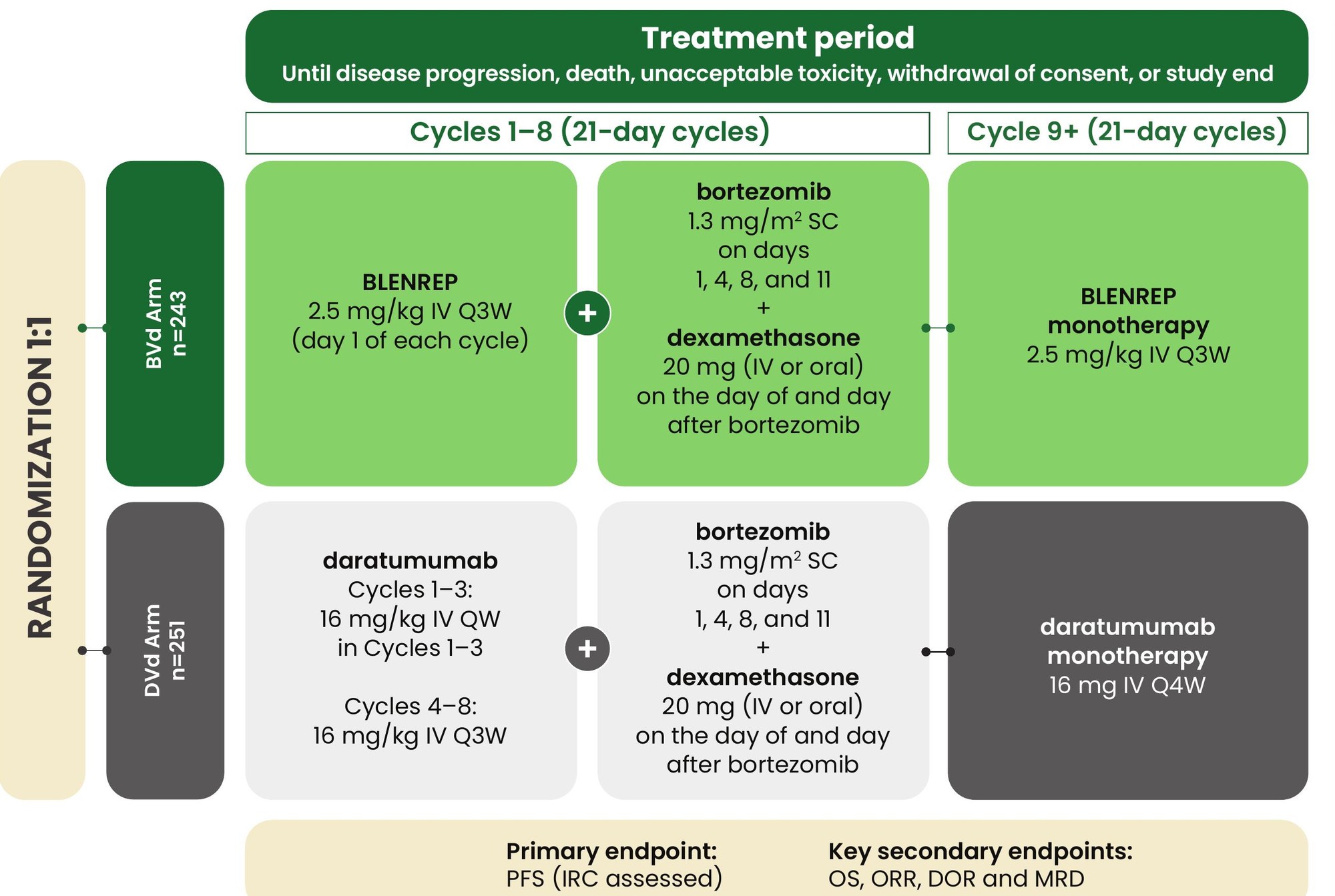
A head-to-head trial designed to study the efficacy of a BLENREP triplet (BVd) vs a daratumumab triplet (DVd) in RRMM
DREAMM-7: A Phase 3, open-label, randomized multicentre study with MM patients who received at least one prior therapy.
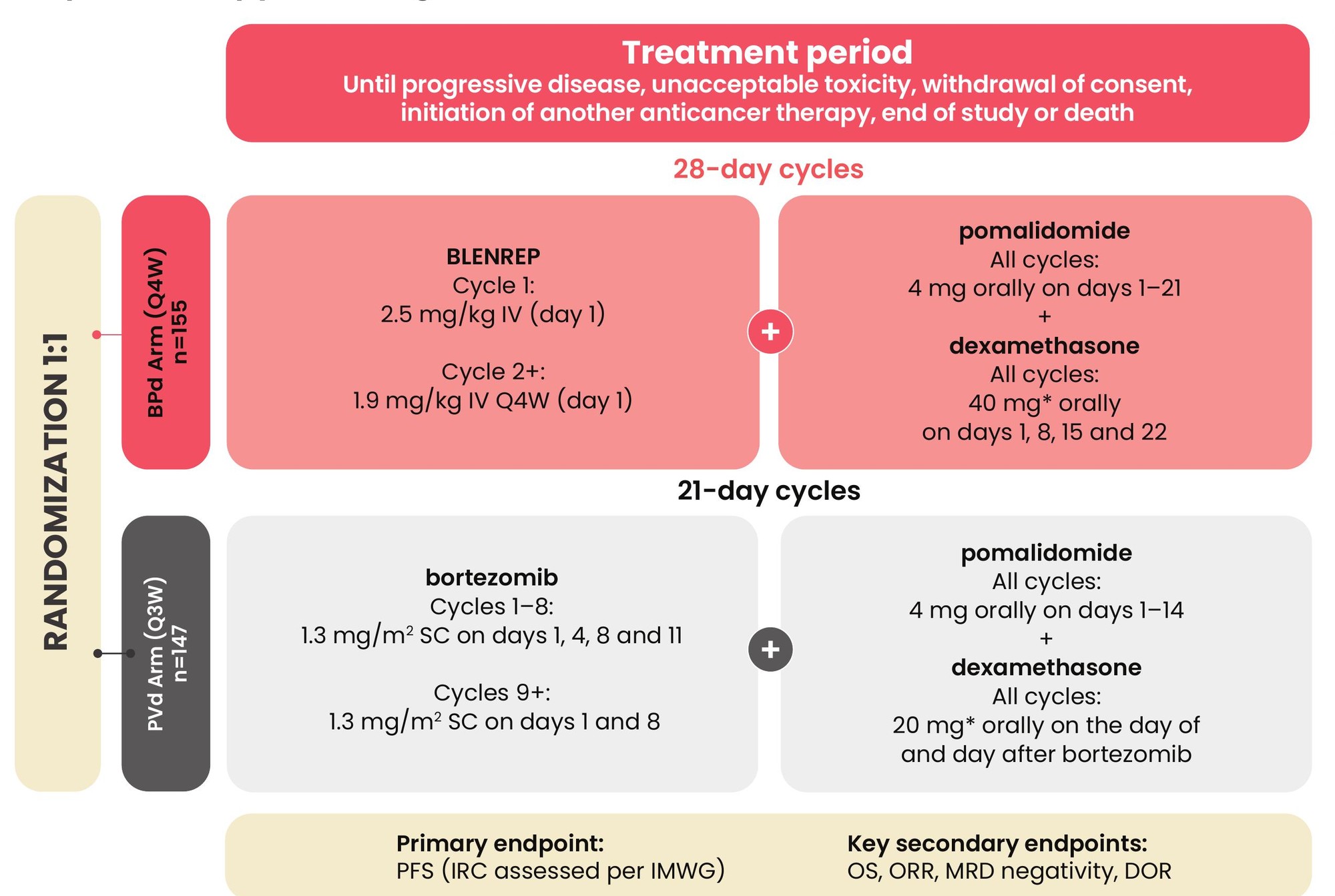
A pivotal Phase 3 trial designed to study the efficacy of a BLENREP triplet (BPd) vs a pomalidomide triplet (PVd) in RRMM
DREAMM-8: A Phase 3, open-label, randomized multicentre study with MM patients who received at least one prior therapy including lenalidomide.
Baseline characteristics
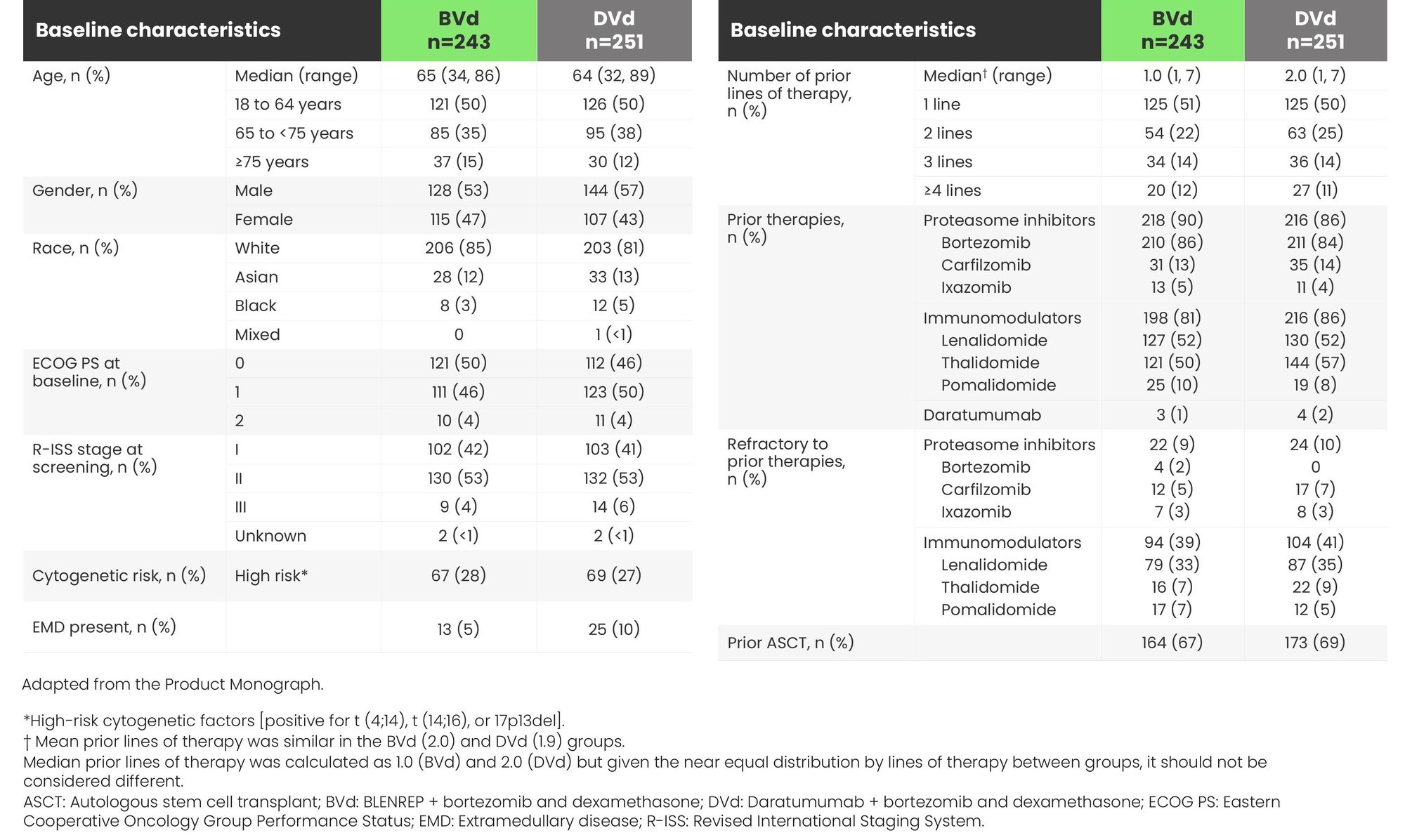
DREAMM-7: baseline characteristics were balanced in both arms
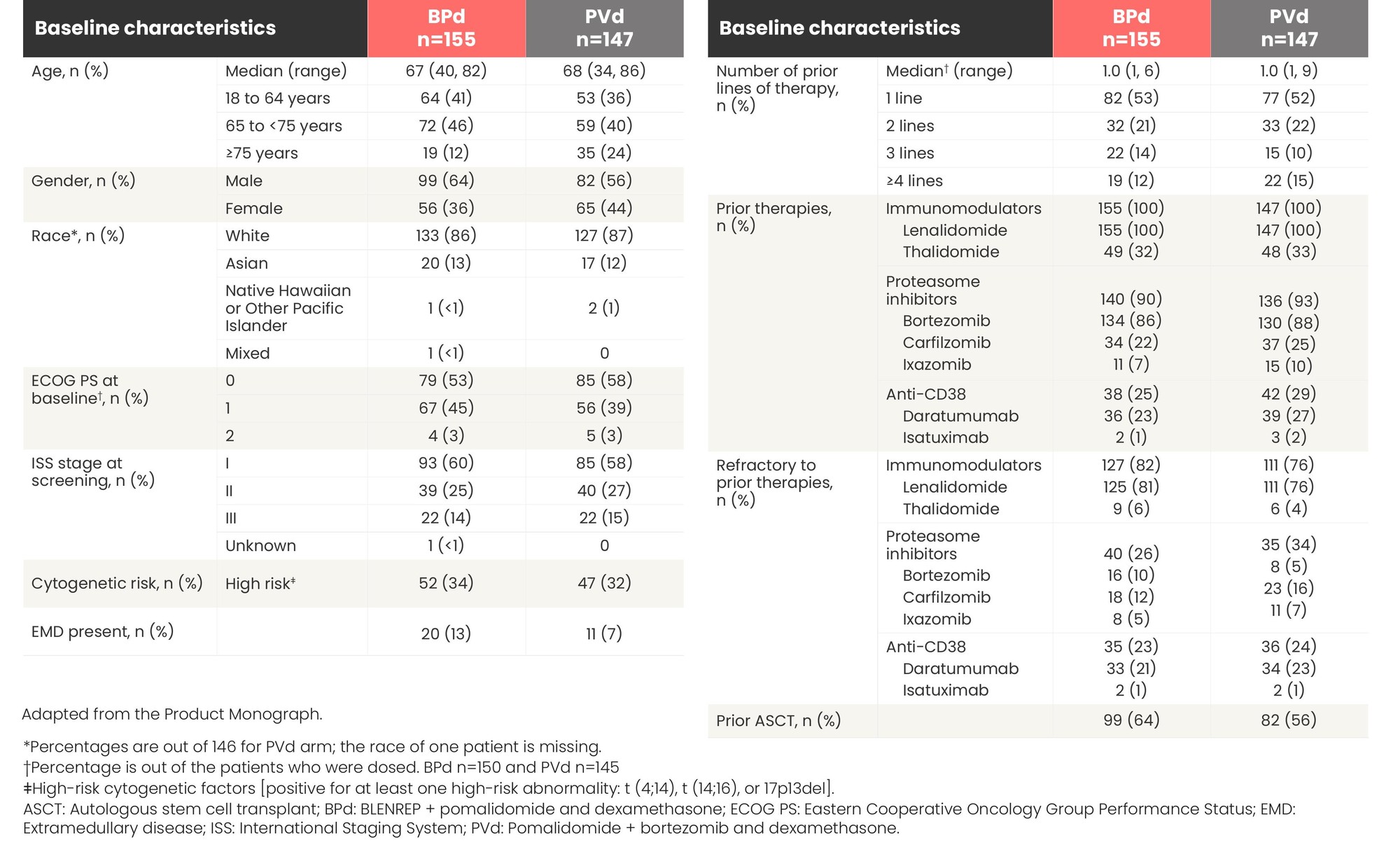
DREAMM-8: baseline characteristics were balanced in both arms
Eligibility criteria
Both DREAMM-7 and DREAMM-8 used the same key eligibility and exclusion criteria.
Key eligibility criteria1:
- A confirmed diagnosis of MM as defined by International Myeloma Working Group (IMWG) criteria
- Previous treatment with at least 1 line of MM therapy
- Documented disease progression during or after their most recent therapy
- ECOG status of 0–2
Key exclusion criteria1:
- Patients refractory or intolerant to daratumumab or bortezomib, or with prior exposure to anti-BCMA therapy
- Patients with current corneal disease (except for mild punctate keratopathy)

