Dose modifications
Modification schedule
BLENREP dose modifications for eye-related side effects in combination with Vd
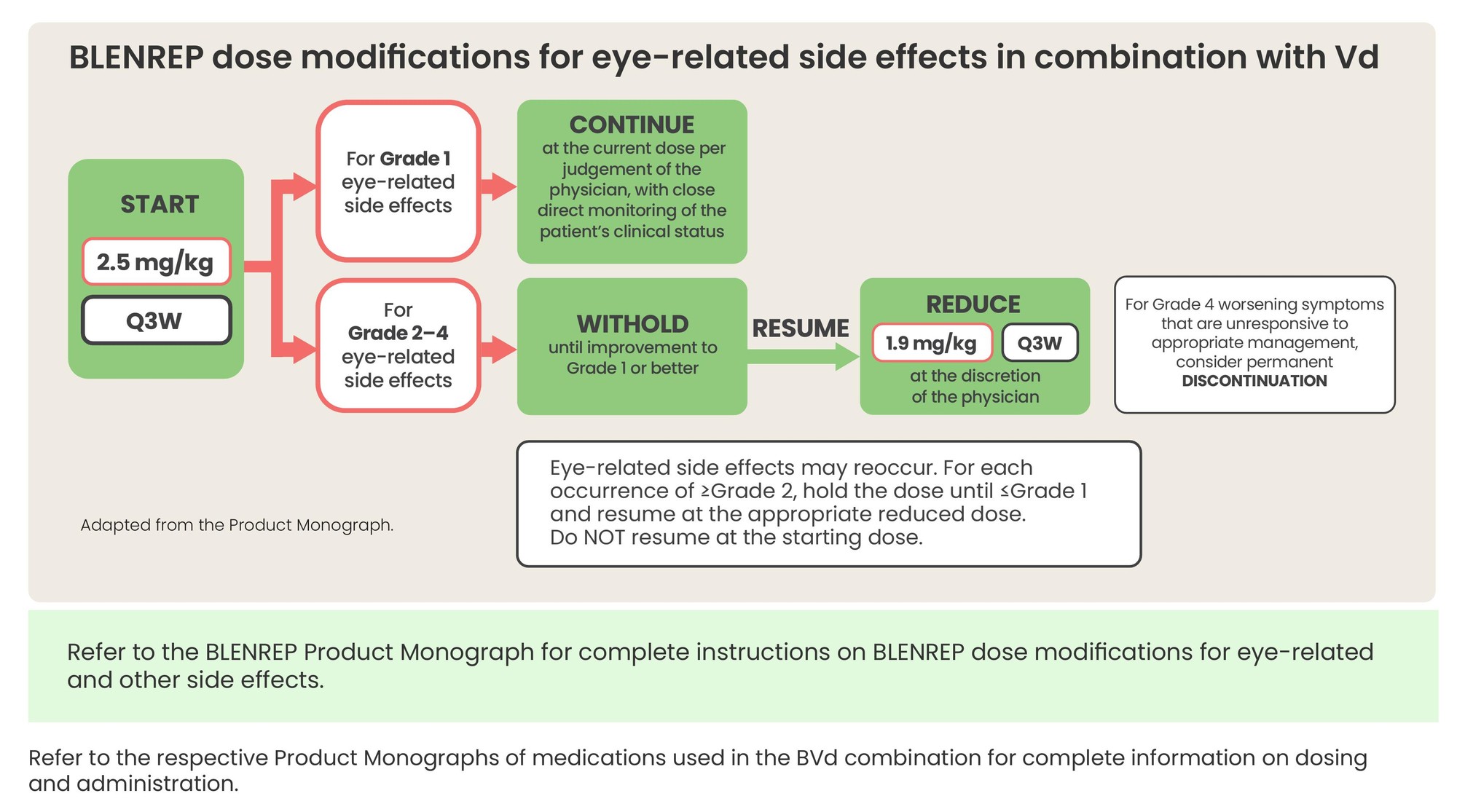
- Refer to the respective Product Monographs of medications used in the BVd combination for complete information on dosing and administration.
*Ocular adverse reaction severity is defined by the most severely affected eye as both eyes may not be affected to the same degree.
BVd: BLENREP + bortezomib and dexamethasone; ERSE: Eye-related side effects; Q3W: Every three weeks; AR: Adverse reaction.
BLENREP dose modifications for eye-related side effects in combination with Pd
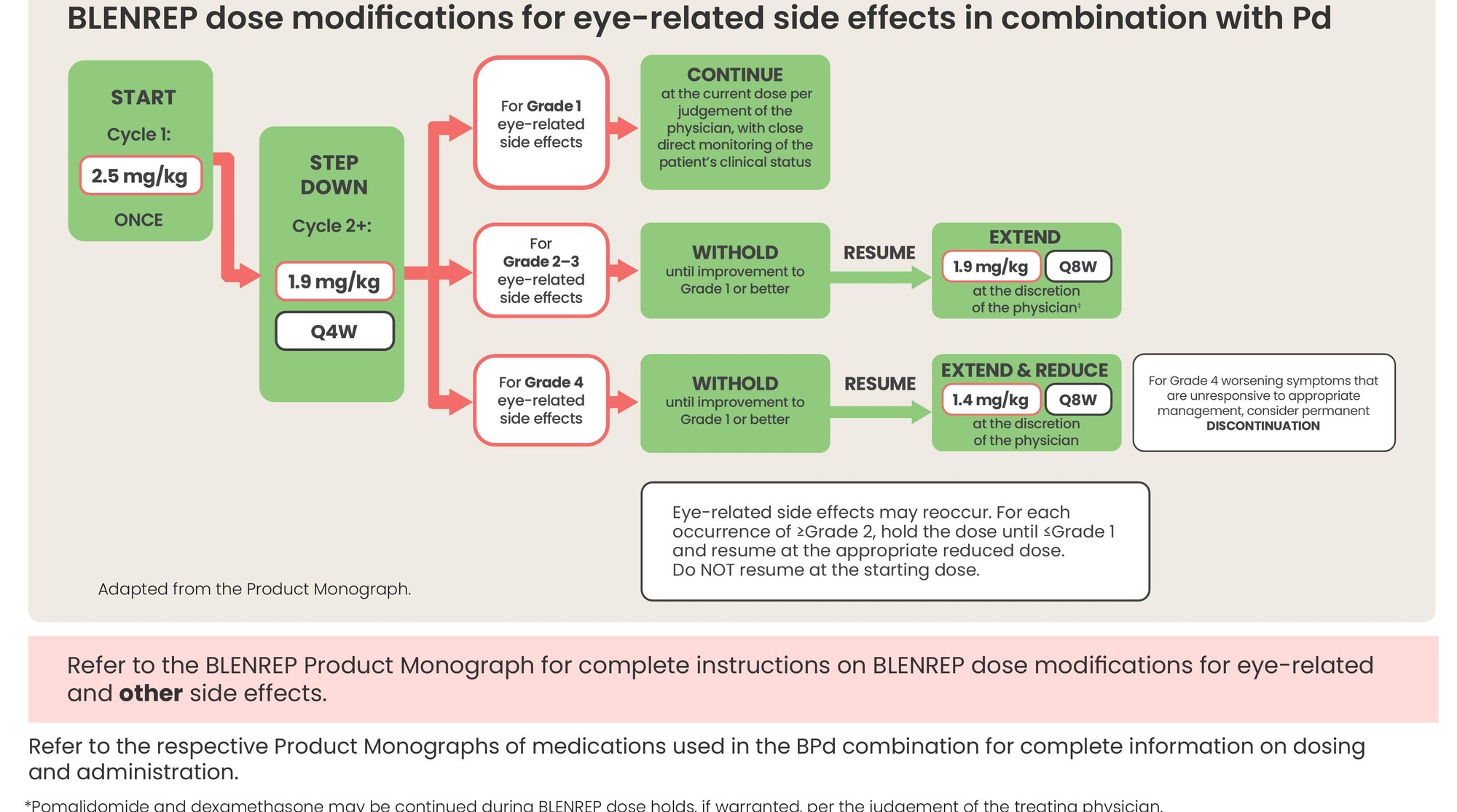
- Refer to the respective Product Monographs of medications used in the BPd combination for complete information on dosing and administration.
*Pomalidomide and dexamethasone may be continued during BLENREP dose holds, if warranted, per the judgement of the treating physician.
†Ocular adverse reaction severity is defined by the most severely affected eye as both eyes may not be affected to the same degree.
‡If toxicity is identified prior to dosing cycle 2 for BPd, dose at 1.9 mg/kg every 4 weeks.
BPd: BLENREP + pomalidomide and dexamethasone; AR: Adverse reaction; ERSE: Eye-related side effects; Q4W: Every four weeks; Q8W: Every eight weeks.
Observed dose modifications
Dose modifications, including delays and reductions, were frequently required to manage adverse reactions, including eye-related side effects
- Among patients who experienced an eye-related side effect, 91% (n=190/209) continued treatment on or after the onset of the first event and received a median of 8 additional doses (range: 1 to 52)
- Permanent discontinuation of any component of BVd due to eye-related side effects, including ocular adverse reactions, visual acuity changes, or corneal examination findings, occurred in 10% of patients
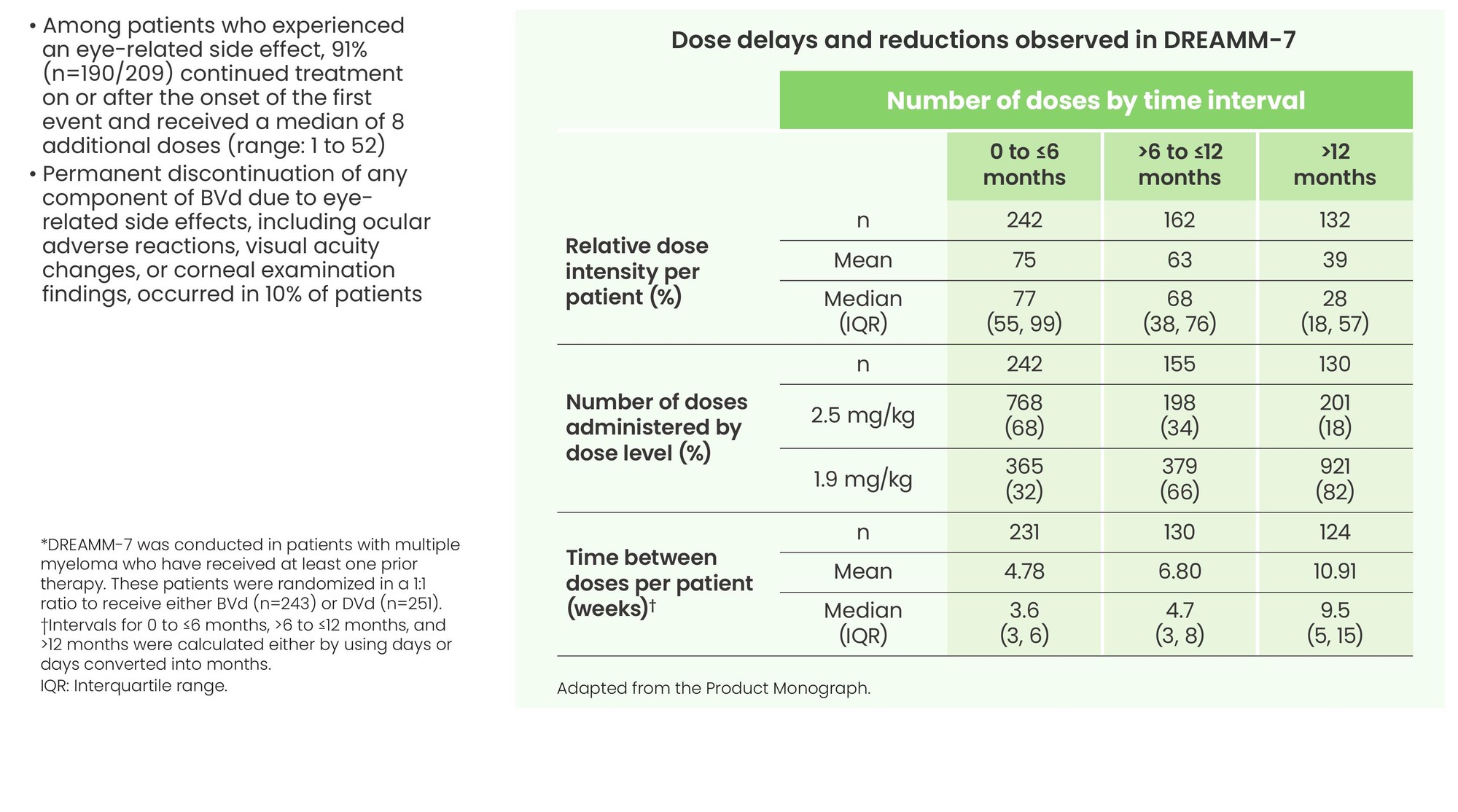
*DREAMM-7 was conducted in patients with multiple myeloma who have received at least one prior therapy. These patients were randomized in a 1:1 ratio to receive either BVd (n=243) or DVd (n=251).
†Intervals for 0 to ≤6 months, >6 to ≤12 months, and >12 months were calculated either by using days or days converted into months.
IQR: Interquartile range.
Dose modifications, including delays and reductions, were frequently required to manage adverse reactions, including eye-related side effects
- Among patients who experienced an eye-related side effect, 92% (n=120/131) continued treatment on or after the onset of the first event and received a median of 5 additional doses (range: 1 to 21)
- Permanent discontinuation of any component of BPd due to eye-related side effects, including ocular adverse reactions, visual acuity changes, or corneal examination findings, occurred in 11% of patients
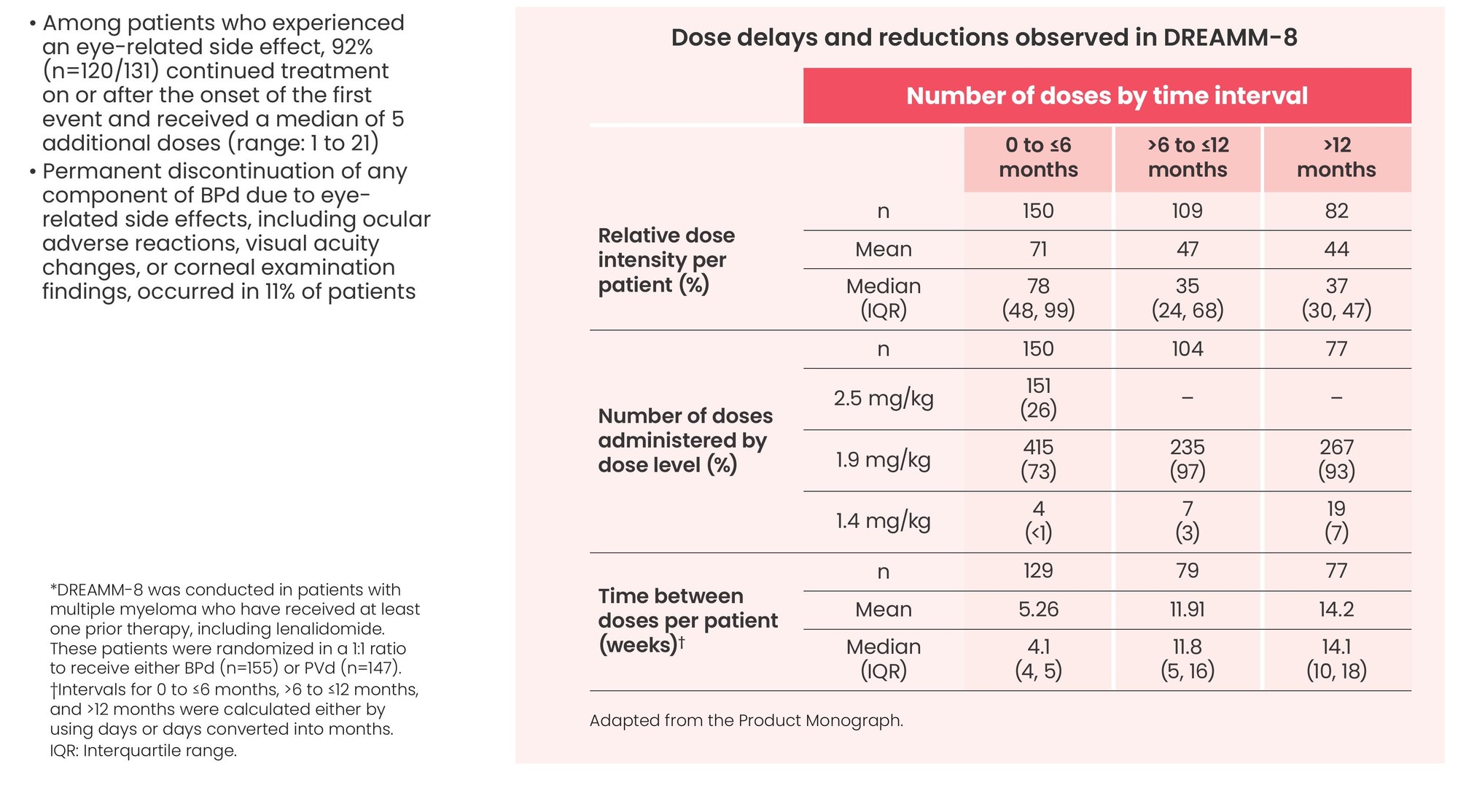
*DREAMM-8 was conducted in patients with multiple myeloma who have received at least one prior therapy, including lenalidomide. These patients were randomized in a 1:1 ratio to receive either BPd (n=155) or PVd (n=147).
†Intervals for 0 to ≤6 months, >6 to ≤12 months, and >12 months were calculated either by using days or days converted into months.
IQR: Interquartile range.

