Efficacy outcomes
Primary endpoint
PFS
At a median follow-up of 28.2 months, BLENREP + Vd significantly improved PFS vs daratumumab + Vd in the ITT population (HR: 0.41; 95% CI: 0.31–0.53; p<0.00001)1,2*
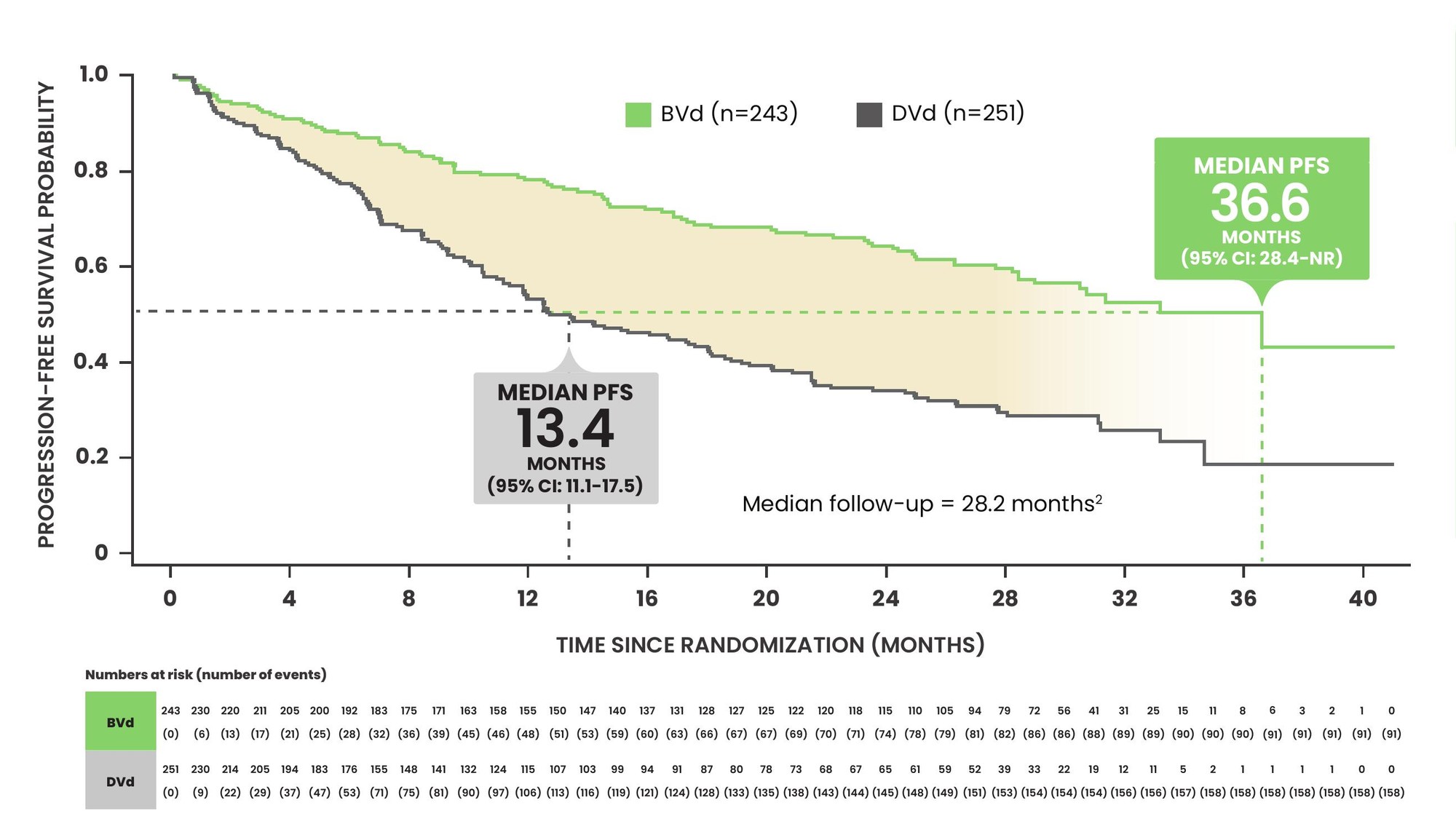
PFS improvement in pre-specified subgroups
The PFS improvement with BLENREP + Vd was consistently observed in all pre-specified subgroups, including patients who1,3:
- Received one prior line of therapy (HR 0.52 [95% CI: 0.36, 0.76])
- Are refractory to lenalidomide (HR 0.37 [95% CI: 0.24, 0.56])
- Have high-risk cytogenetics (HR 0.36 [95% CI: 0.22, 0.58])
At a median follow-up of 21.8 months, BLENREP + Pd significantly improved PFS vs pomalidomide + Vd in the ITT population (HR: 0.52; 95% CI: 0.37–0.73; p<0.001)1*
Median PFS was NR for BLENREP + Pd vs 12.7 months for pomalidomide + Vd1*
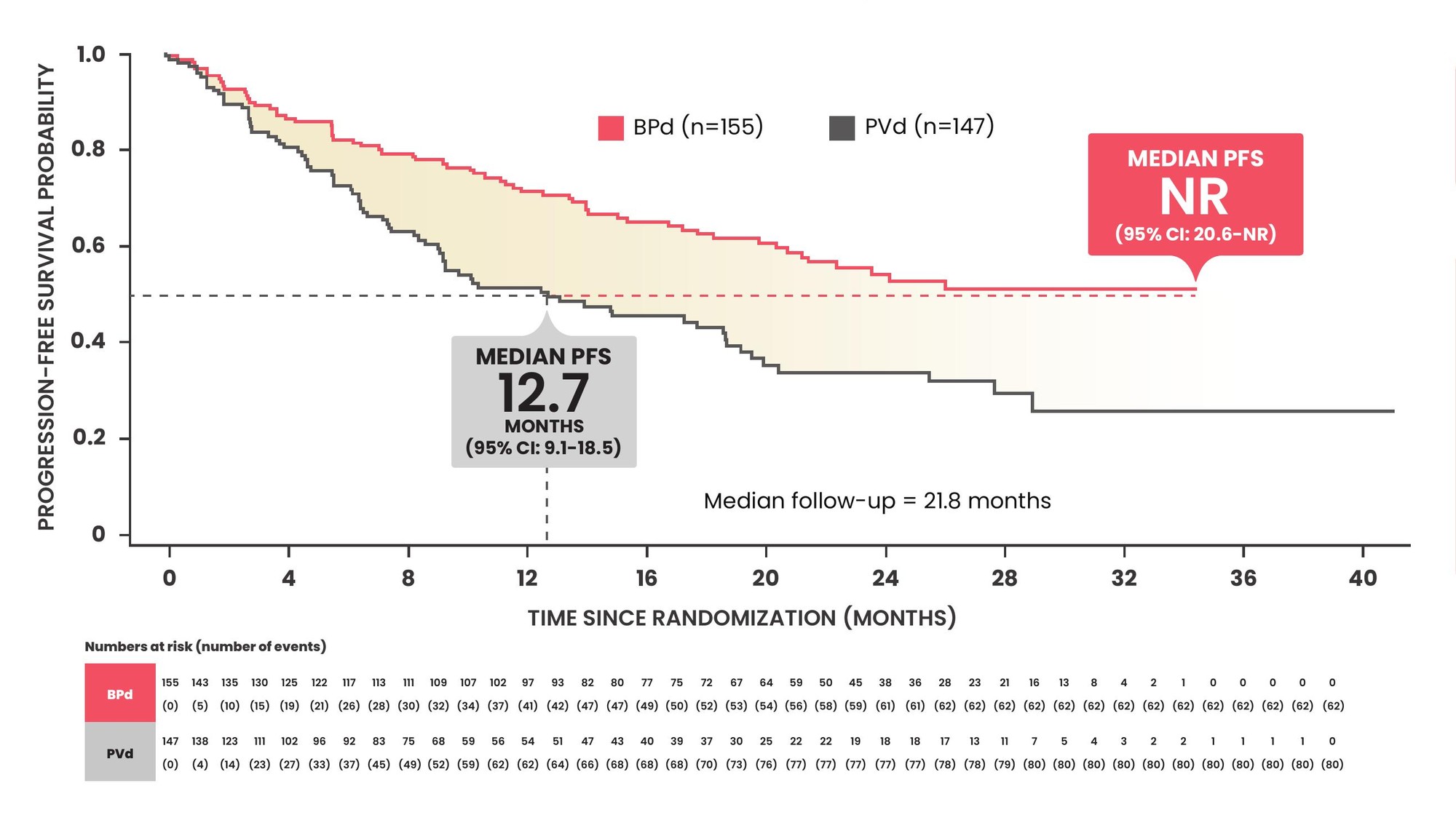
PFS improvement in pre-specified subgroups
The PFS improvement with BLENREP + Pd was consistently observed in all pre-specified subgroups, including patients who1,3:
- Have not received a prior anti-CD38 treatment (HR 0.48 [95% CI: 0.32, 0.73])
- Received 1 prior line of therapy (HR 0.52 [95% CI: 0.31, 0.88])
- Have high-risk cytogenetics (HR 0.57 [95% CI: 0.34, 0.95])
- Are refractory to lenalidomide (HR 0.45 [95% CI: 0.31, 0.65])
- Are refractory to anti-CD38 agents (HR 0.65 [95% CI: 0.36, 1.18])
Secondary endpoint
OS
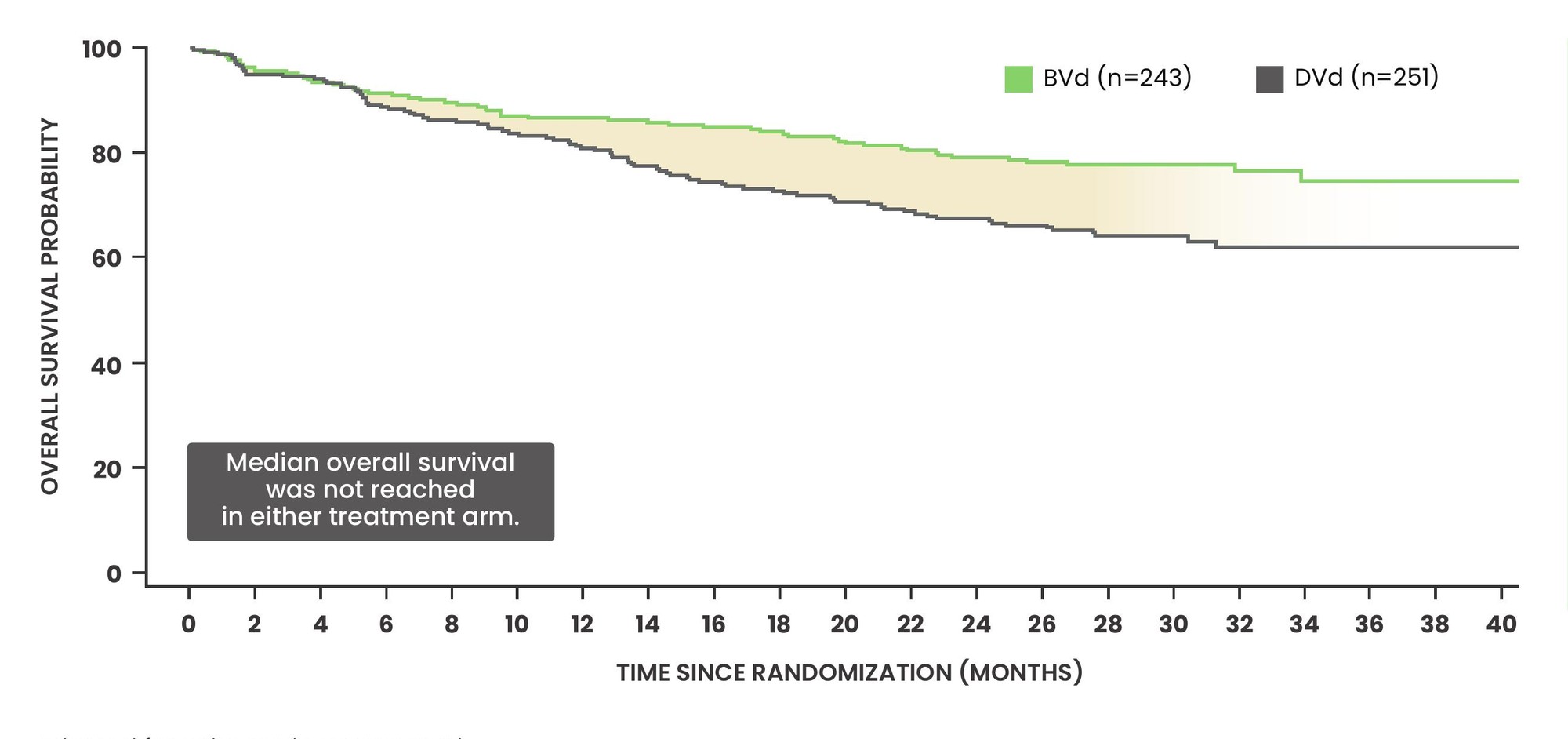
First interim analysis of overall survival (OS) at 18 months with BLENREP + Vd vs daratumumab + Vd1
- 43% reduction in the risk of death was observed in favour of BVd (HR: 0.57; 95% CI: 0.40–0.80) 54/243 vs 87/251
- OS data are immature as only 40% of events have occurred to date (n=141/355 events)
- Follow-up for overall survival is ongoing
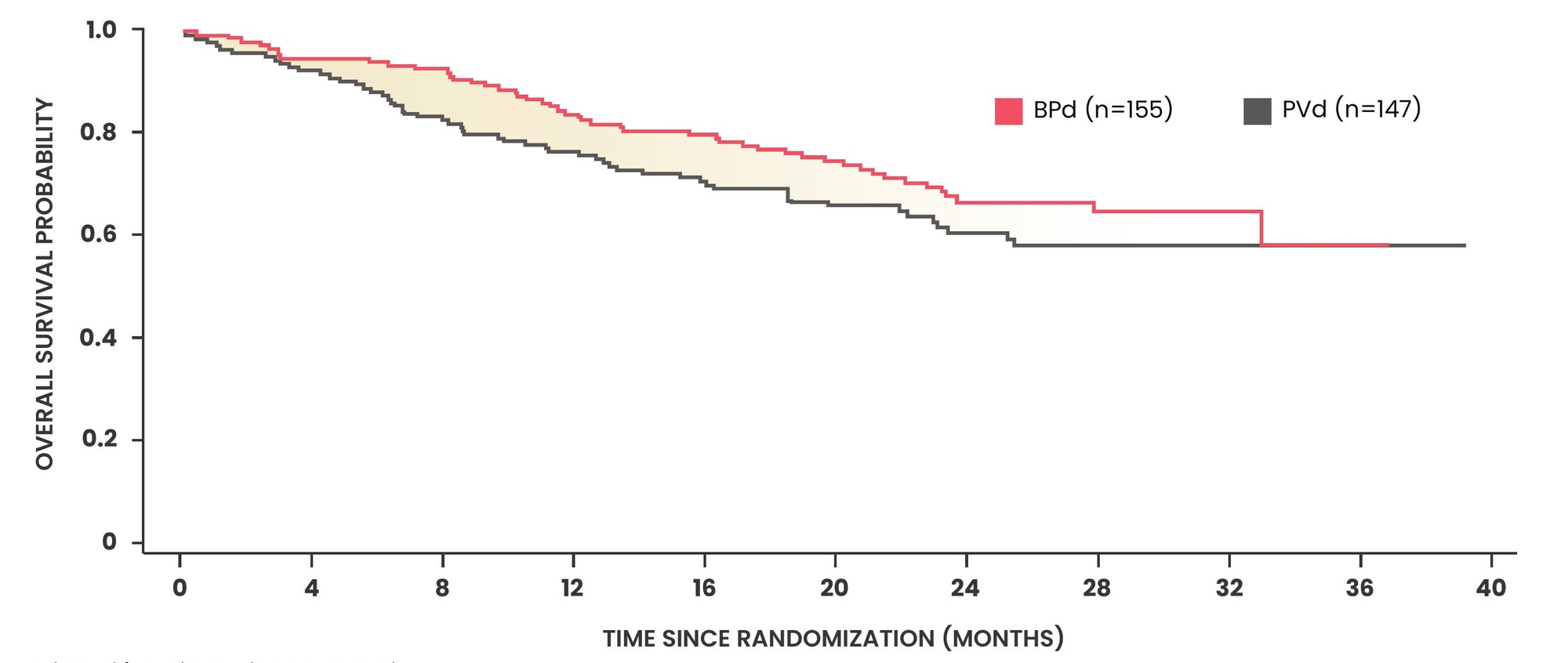
First interim analysis of overall survival (OS) with BLENREP + Pd vs pomalidomide + Vd1,2
- The estimated OS HR was 0.77 (95% CI: 0.53–1.14)
- OS data are immature as only 48% of events have occurred to date (n=105/217 events)
- Follow-up for overall survival is ongoing
Secondary endpoint
ORR
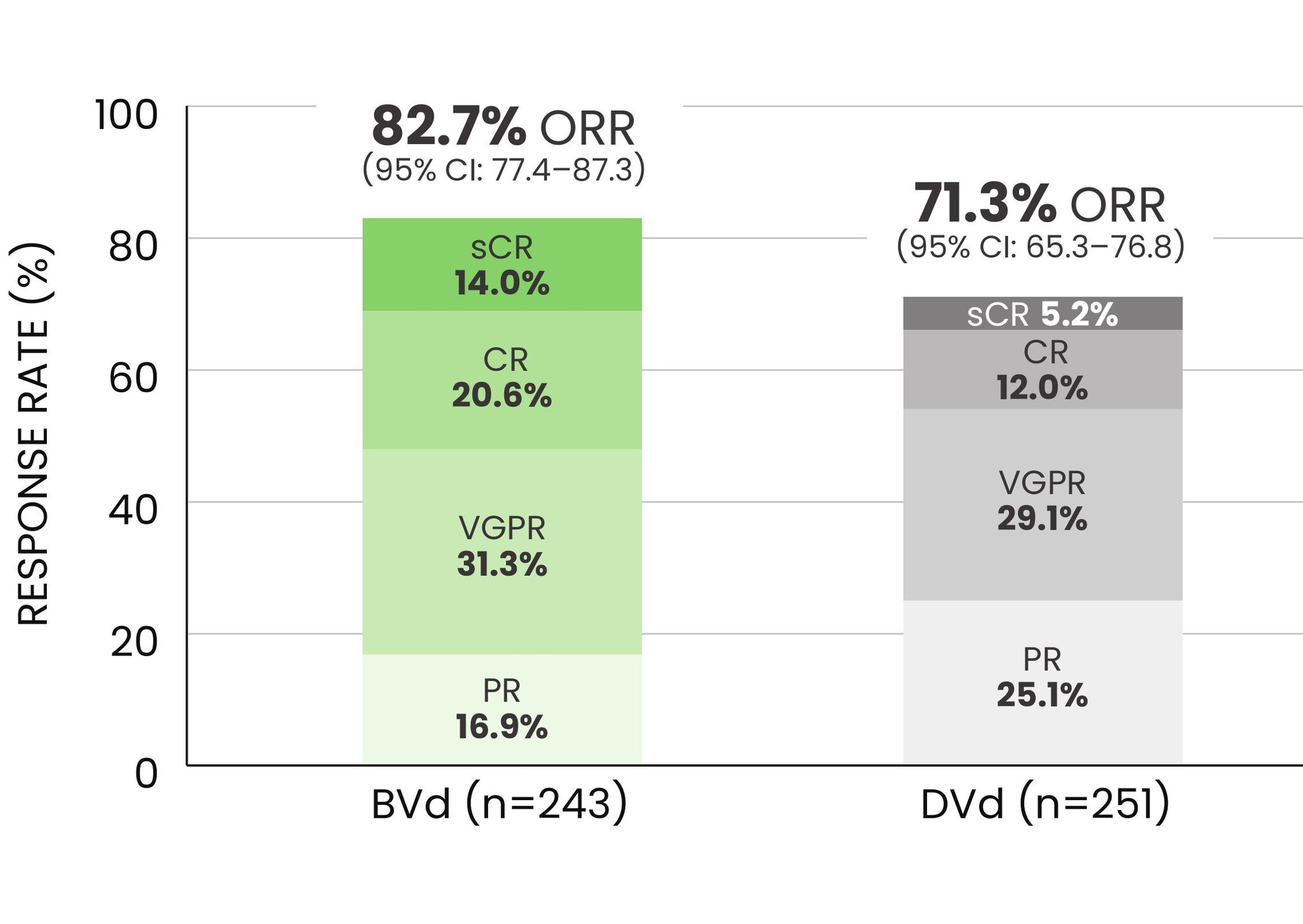
The overall response rate (ORR) was 82.7% for patients treated with BLENREP + Vd vs 71.3% with daratumumab + Vd*
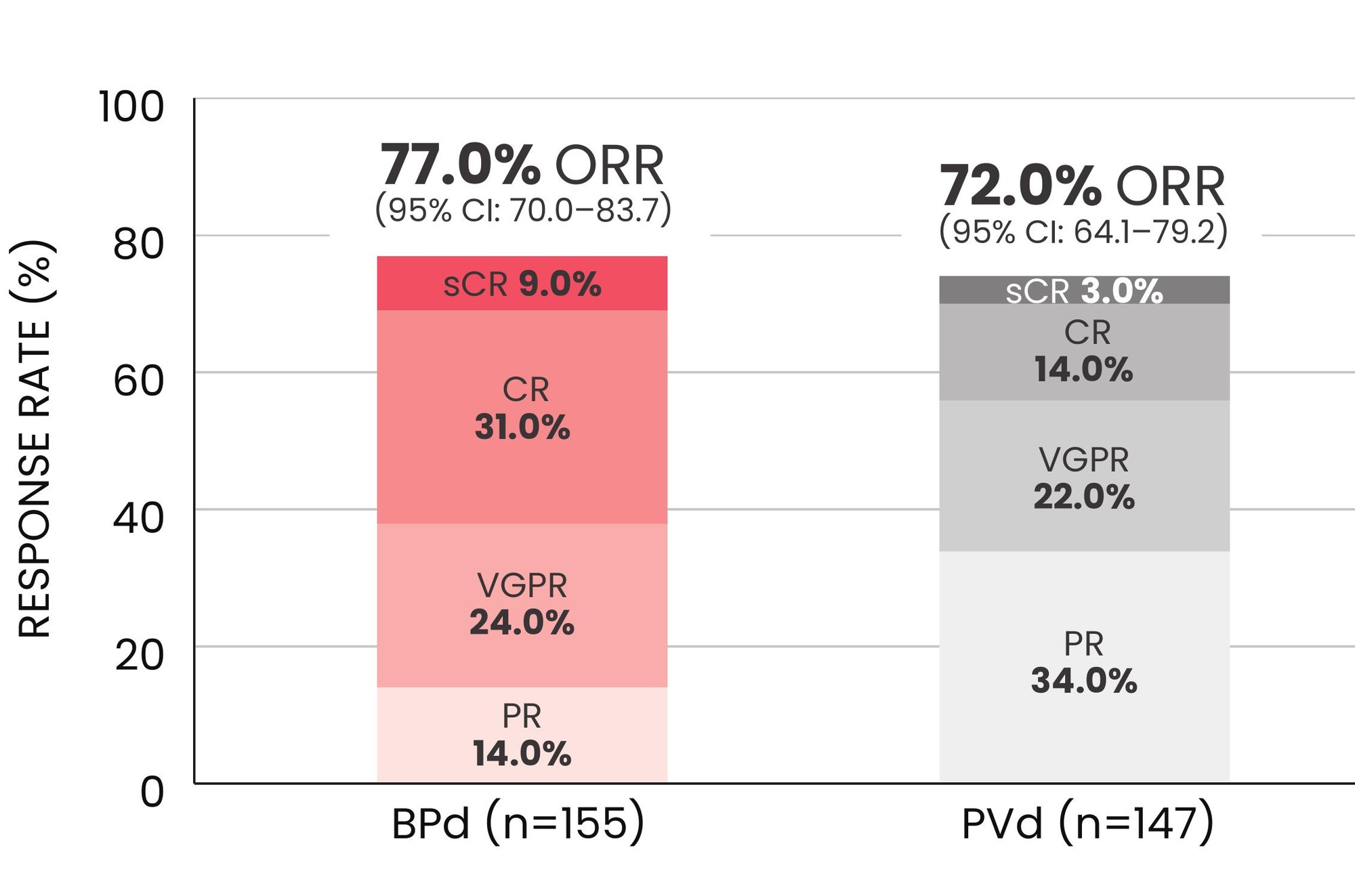
The overall response rate (ORR) was 77% for patients treated with BLENREP + Pd vs 72% with pomalidomide + Vd*
Secondary endpoint
MRD
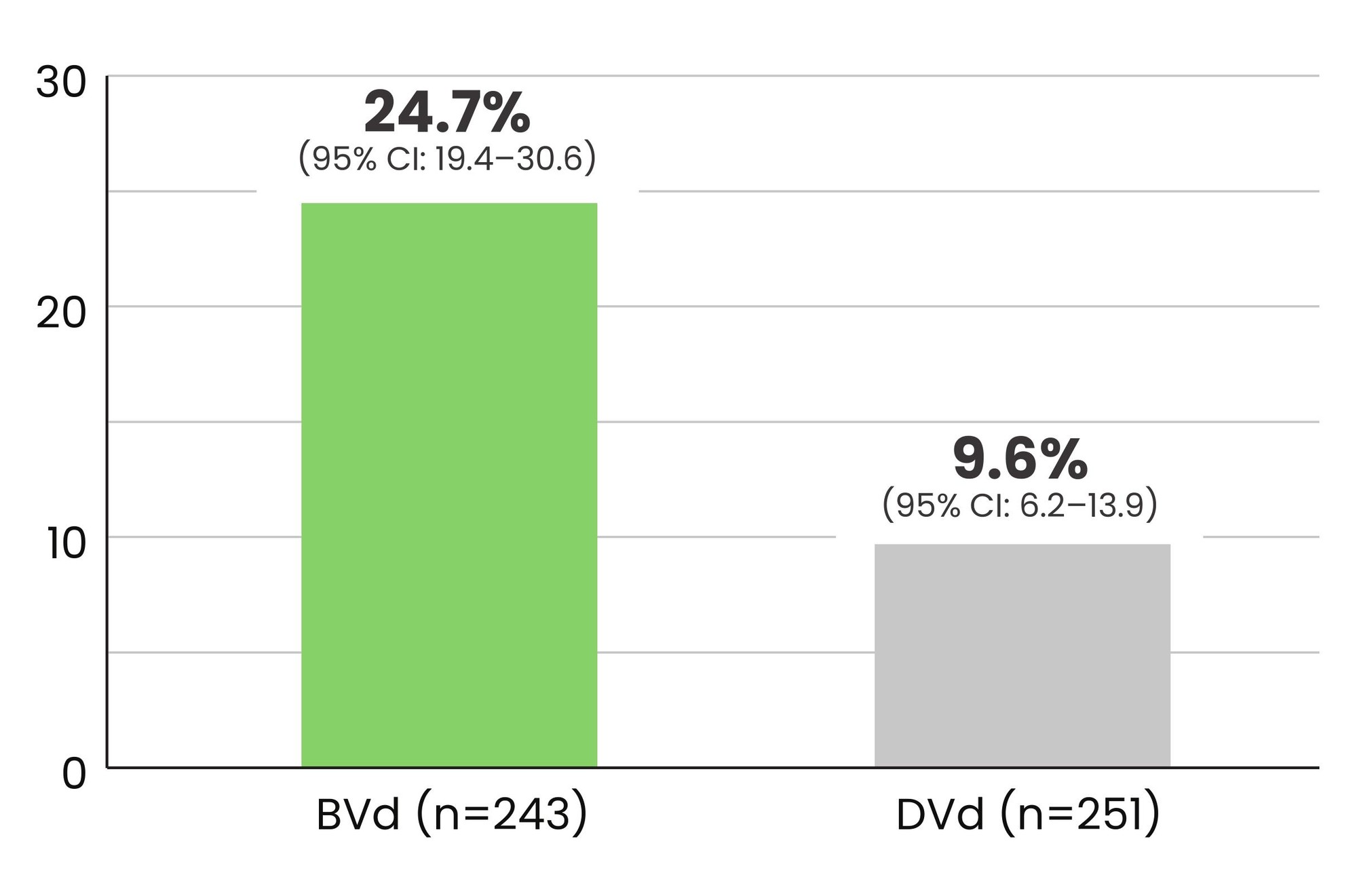
24.7% of patients receiving BLENREP + Vd achieved MRD negativity vs 9.6% of patients receiving daratumumab + Vd1,2*†
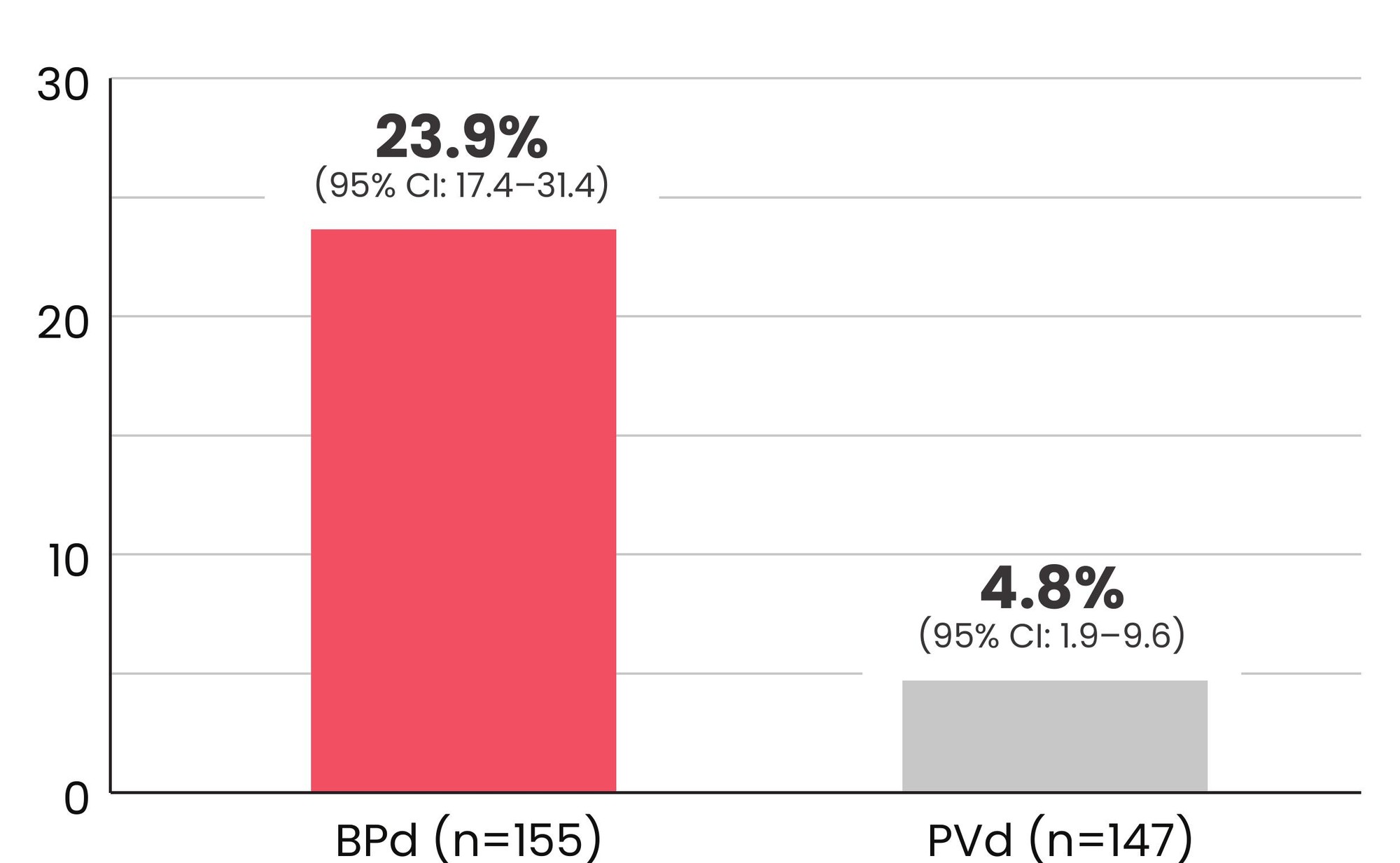
23.9% of patients receiving BLENREP + Pd achieved MRD negativity vs 4.8% of patients receiving pomalidomide + Vd1,2*†
*Evaluated by a blinded Independent Review Committee (IRC) based on the International Myeloma Working Group (IMWG) criteria for multiple myeloma.
†For patients with a complete response or better.
‡Assessed by next-generation sequencing (NGS) at the 10⁻⁵ threshold.
BVd: BLENREP + bortezomib and dexamethasone; BPd: BLENREP + pomalidomide and dexamethasone; CI: confidence interval; DOR: duration of response; DVd: daratumumab + bortezomib and dexamethasone; HR: hazard ratio; ITT: intent-to-treat; MRD: minimal residual disease; NR: not reached; ORR: overall response rate; OS: overall survival; PFS: progression-free survival; PR: partial response; PVd: pomalidomide + bortezomib and dexamethasone; Pd: pomalidomide and dexamethasone; Vd: bortezomib and dexamethasone.

